Asymptotic perturbation and sensitivity analysis of malaria transmission in Nigeria: a mathematical model
Keywords:
Malaria transmission, Perturbation method, Sensitivity analysis, Basic reproduction numberAbstract
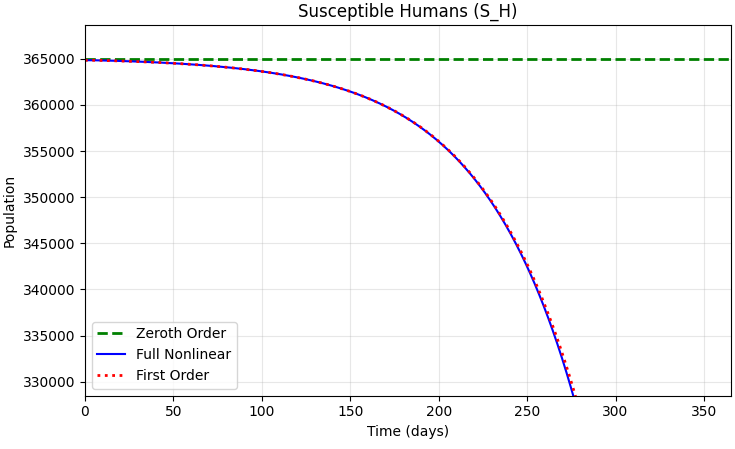
This article analyzes malaria dynamics among high-risk groups in Nigeria, with the human population divided into five compartments and the mosquito population divided into two compartments. The resulting system of nonlinear ordinary differential equations is examined by asymptotic perturbation and sensitivity-analysis methods. The perturbation analysis describes the behavior of the system at the malaria-free equilibrium (MFE) and the effect of a small perturbation around that state. The first-order perturbation solution indicates exponential growth in disease prevalence, showing that the MFE is unstable when infection is introduced. The sensitivity analysis shows that preventing mosquitoes from surviving long enough to become infectious is the most effective way to reduce the malaria transmission cycle. The infected adult male compartment, IM, also acts as an important long-term reservoir and is the second most influential factor in the model. The estimated basic reproduction number satisfies R0 > 1, indicating a high potential for malaria spread and the need for effective control measures.
Published
How to Cite
Issue
Section
Copyright (c) 2026 Sunday N. Aloke, Henry O. Adagba, Uchenna E. Michael, Okorie Nwite, Theresa E. Efor, Aloysius N. Ezaka, Chika Agha

This work is licensed under a Creative Commons Attribution 4.0 International License.







